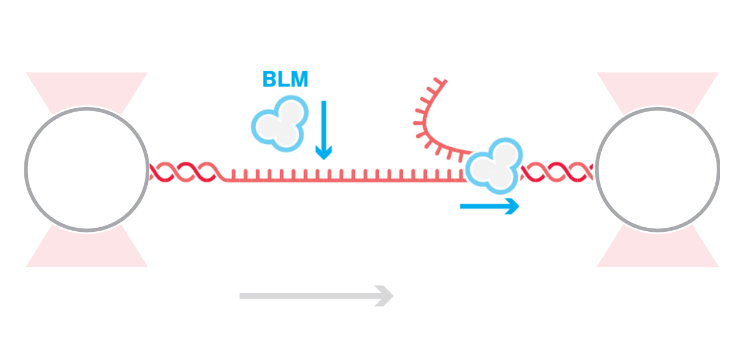
A study published in eLife shows how replication protein A (RPA) activates helicase Bloom syndrome protein (BLM) to unwind double-stranded DNA (dsDNA) bidirectionally from nicks. The C-Trap® enabled the researchers to simultaneously measure and visualize the DNA–protein interactions in real time.
The findings highlight previously unknown mechanisms of BLM that can aid in the development of therapies against heritable diseases associated with BLM and related helicases.
Congratulations to Dr. Bo Sun at ShanghaiTech University, his lab, and all the authors involved in this work!
Tethering a nicked dsDNA between two optically trapped beads enabled the team to record BLM-induced unwinding. By simultaneously measuring the lengthening associated with unwinding and following the dsDNA-specific fluorescent Sytox Orange, they could study how the helicase processed the DNA in different conditions and at different stretching forces.
In the absence of RPA, BLM could unwind the dsDNA unidirectionally, and only upon DNA stretching and at high concentrations of the helicase. However, the addition of free RPA in the solution induced bidirectional DNA unwinding at lower stretching forces and low BLM concentrations. Of note, the researchers found that the cooperation between RPA and BLM to facilitate bidirectional unwinding was independent of intermolecular interactions.
The findings offer a further mechanistic understanding of the cellular responses to replication stress and DNA damage, which can aid the development of future therapeutic strategies.
“[…] our experimental approach combining optical tweezers with fluorescence microscopy has shed new light on the effect of RPA on BLM’s ability to catalyze dsDNA unwinding,” the authors concluded, “This is the first observation, to our knowledge, of helicase bidirectional unwinding from a nick.”
For more information, read the full article published in eLife and entitled “Human RPA activates BLM’s bidirectional DNA unwinding from a nick”.
Are you interested in using dynamic single-molecule tools like the C-Trap® for your research? Please feel free to contact us for more information, a demo, or a quote.