A recent study in the Biophysical Journal shows how a Dutch research group measured and visualized dynamic interactions between membranes in the presence of Ca2+ sensors and synaptic inducers Syt1 and Doc2b. With the combined optical tweezers and confocal fluorescence microscopy configuration of the C-Trap®, the team revealed Ca2+ sensor-dependent mechanisms in membrane remodeling.
The study provides new evidence that clarifies the required features of Ca2+ sensors when inducing membrane fusion, a process essential for neurotransmitter release.
Congratulations to Prof. Gijs Wuite, his lab, and all the authors involved in this work!
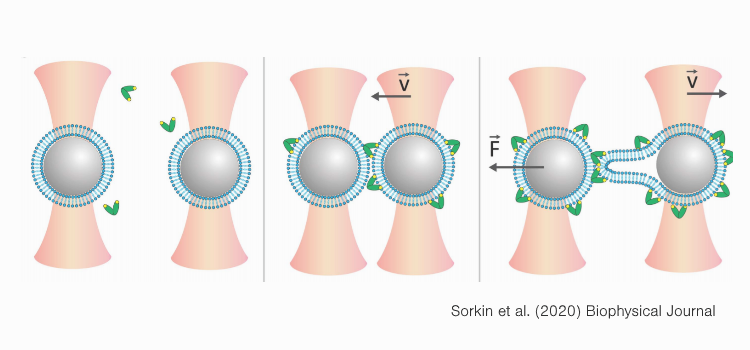
Sorkin et al. used a cell-free model to assess properties associated with membrane–membrane interactions induced by Syt1 or Doc2B. By bringing phospholipid bilayer-coated beads into contact and subsequently pulling them apart, they could measure membrane–membrane interaction probabilities and strength in the presence of these proteins. On top of that, they could monitor retraction events in real time through live confocal imaging to investigate the binding properties of the respective Ca2+ sensors.
The researchers found that Syt1 and Ca2+ levels independently determined the probability and strength of membrane–membrane interactions. These results indicate that the approach serves to detect membrane–membrane interactions in the presence of Ca2+ sensors.
Fluorescently labeling the respective proteins and following them in real time enabled the team to validate membrane fusion mechanisms and differentiate between hemifusion or protein bridging events. The results indicated that both proteins induce hemifusion, albeit Syt1 only at higher concentrations – lower concentrations of Syt1 induced bridging.
The study demonstrates how combining optical tweezers and live imaging in a cell-free model offers functional insights into membrane fusion that are unattainable by existing approaches.
For more information, you can read the full article entitled “Synaptotagmin-1 and Doc2b exhibit distinct membrane remodeling mechanisms” in the Biophysical Journal.
Are you interested in using dynamic single-molecule tools like the C-Trap® for your research? Please feel free to contact us for more information, a demo, or a quote.